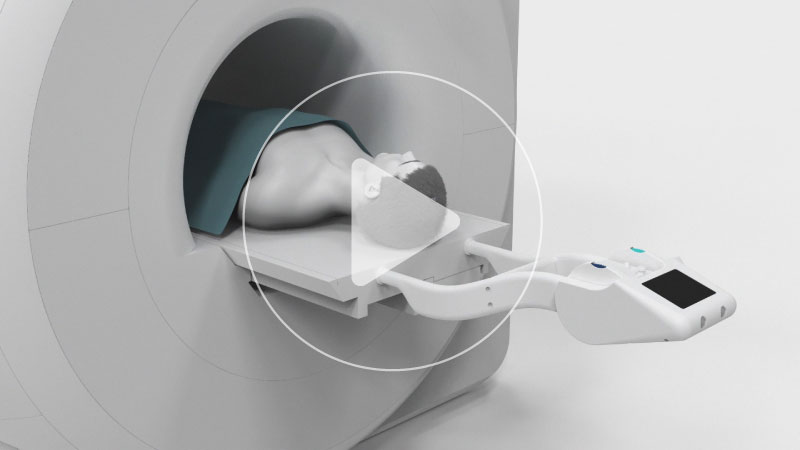
The NeuroBlate® System is the only minimally invasive surgical choice for robotic, cooled laser interstitial thermotherapy (LITT) that uses MR-guided surgical ablation technology designed specifically for use in the brain. NeuroBlate provides precise and maximal brain lesion ablation for adult and pediatric patients without the invasiveness of an open neurosurgical procedure.
WHERE NEUROBLATE IS USED

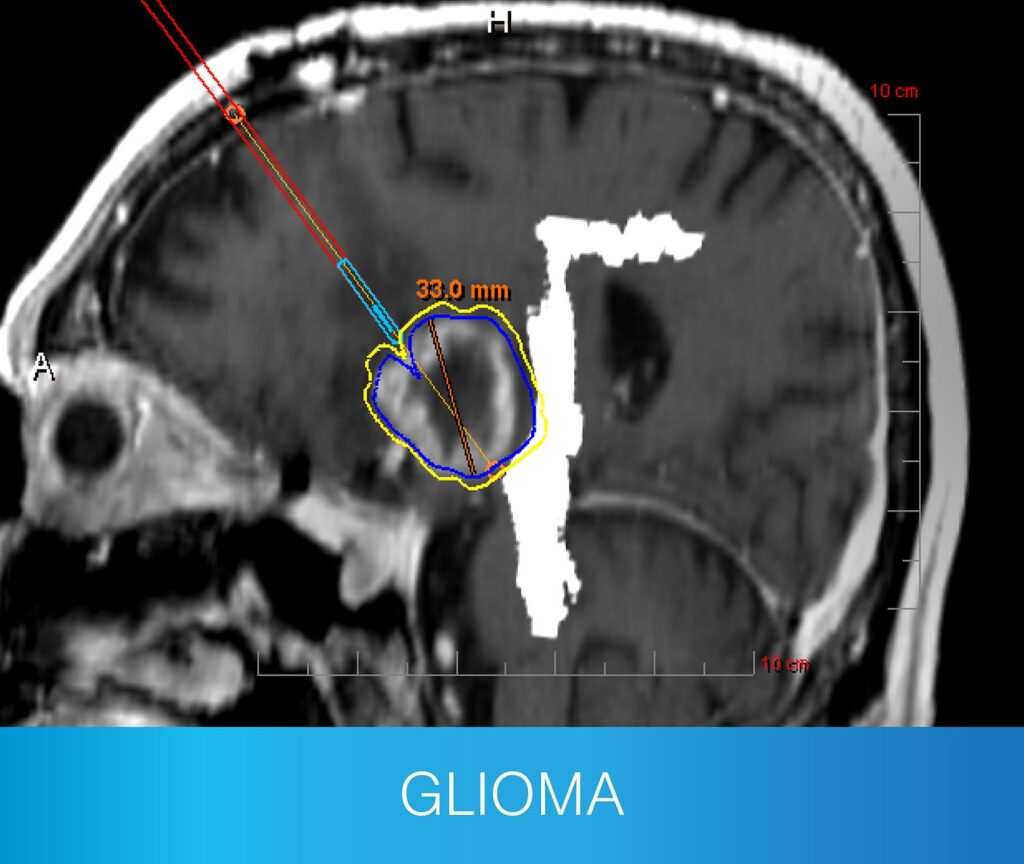
- Newly diagnosed and recurrent primary tumors
- Recurrent metastatic tumors and post-SRS progression
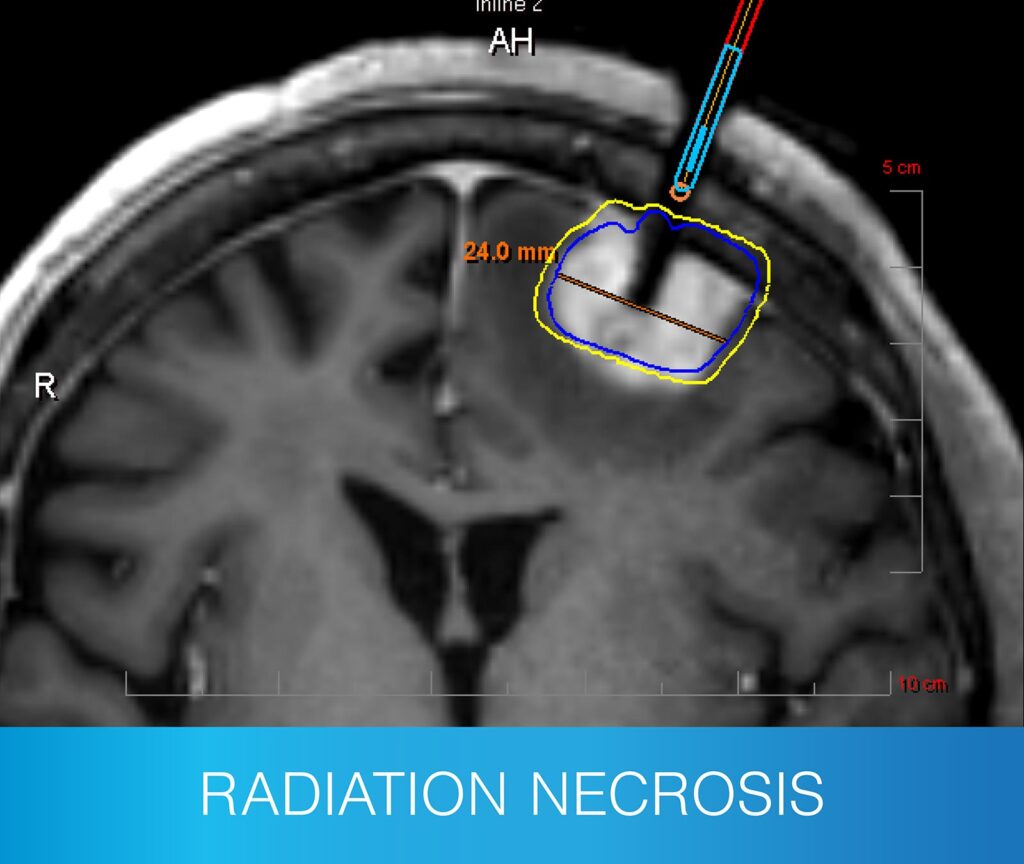
- Radiation necrosis
- Patients with previous interventions
- Deep-seated and difficult to access tumors
- Fragile patients

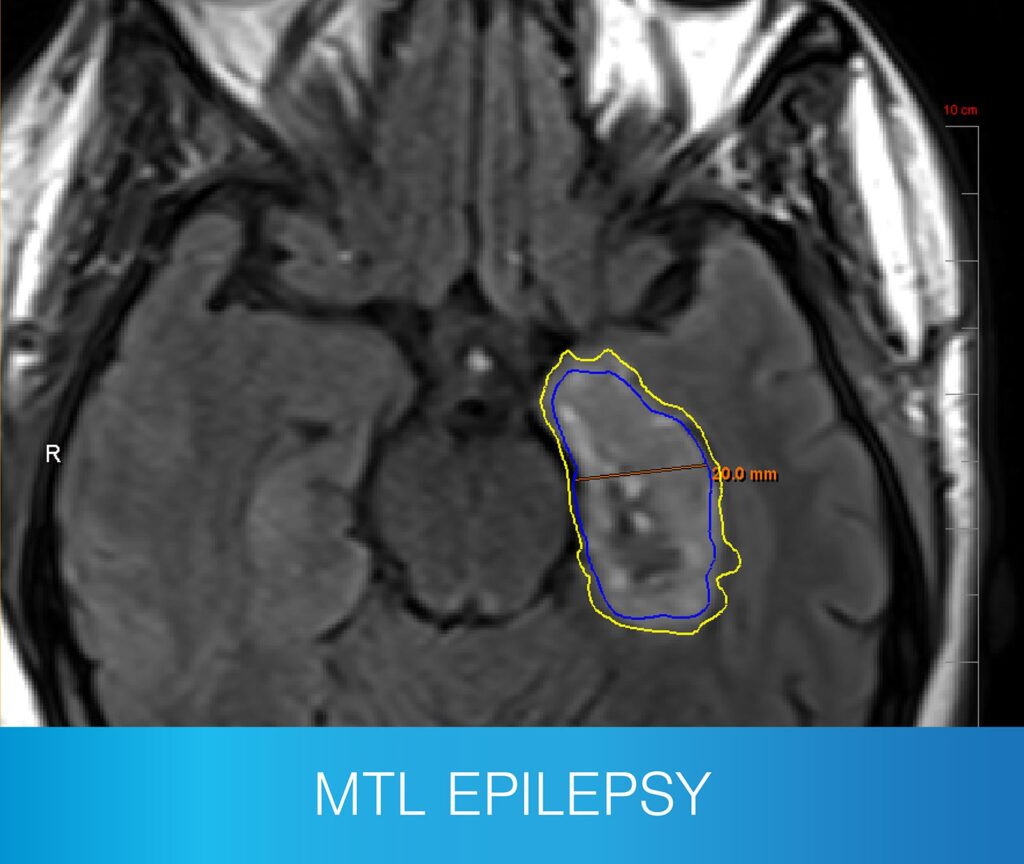
- Mesial temporal lobe ablation
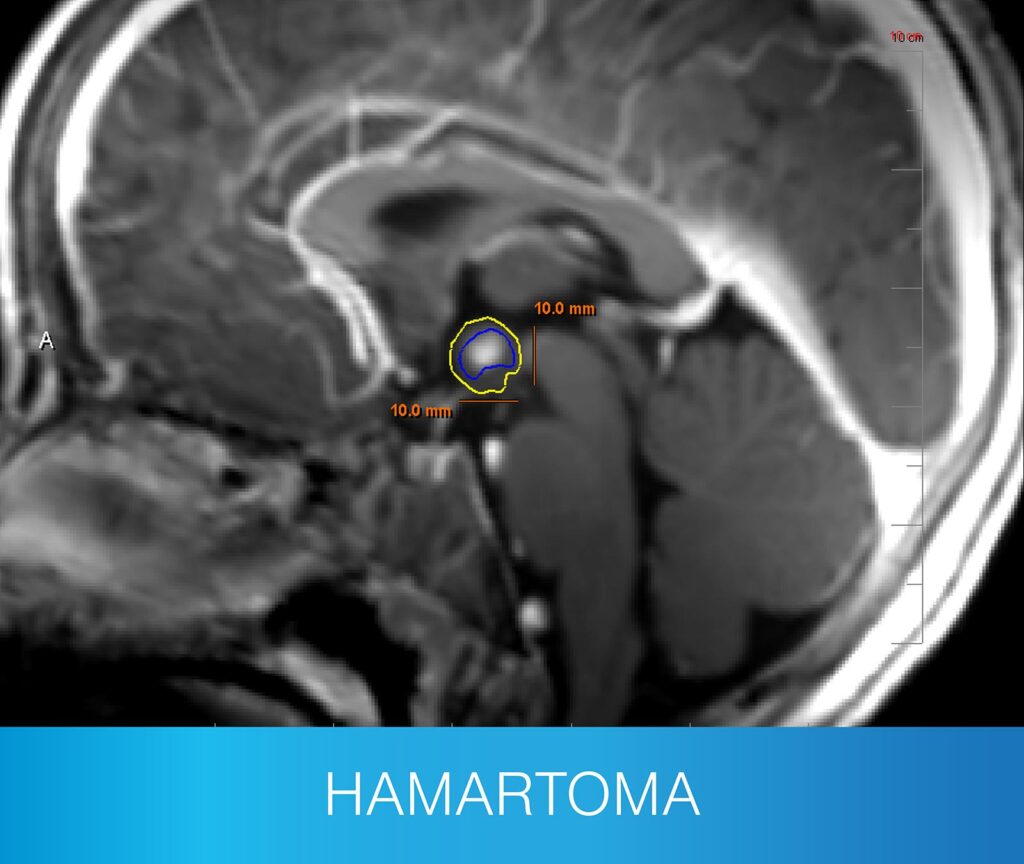
- A range of extratemporal lesions, including hypothalamic hamartoma and corpus callosotomy
- Patients that have trialed more than two drug regimens and are not seizure-free
- Those seeking an intervention without the associated risks of craniotomy
EXPLORE NEUROBLATE LITT
PROVEN
VERSATILITY
+ SAFETY
- Only Monteris offers both FullFire® diffuse and SideFire® directional laser probes – giving surgeons unparalleled versatility to tailor their approach for each unique lesion.
- Proven safe and reliable, NeuroBlate maximizes efficiency, enabling multiple trajectories with a single probe in the same procedure.
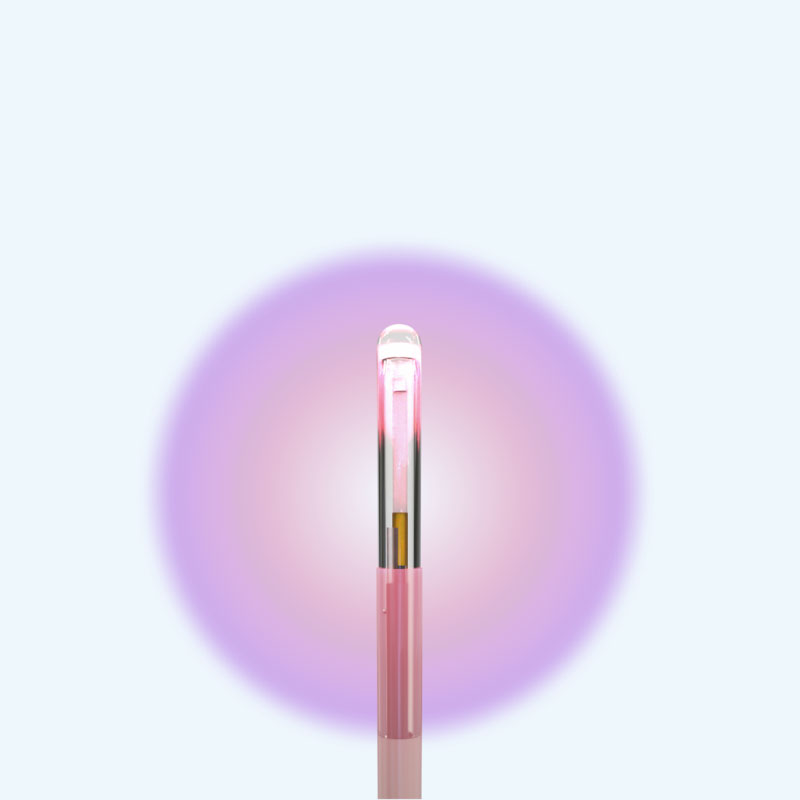
NB3™ FullFire® 1.6mm
Laser Probe

FullFire® 3.3mm
Laser Probe
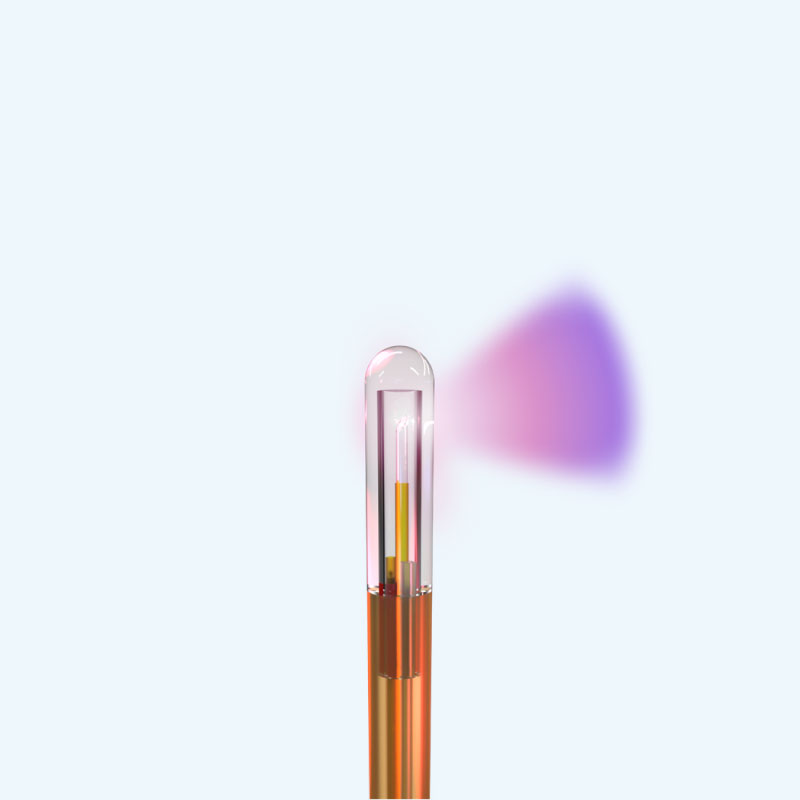
SideFire® 3.3mm
Laser Probe

KNOWN FOR
SUPERIOR
VISUALIZATION
Monteris continuously leads with surgeons’ preferred visualization platform: NeuroBlate® Fusion‑S™ Software — designed for safety and precision.
- Trusted by Neurosurgeons: Fusion-S supports NeuroBlate procedures with tools designed for safety and precision.
- Clear 3D Imaging: Shows detailed views of the brain and heat patterns during treatment to guide the procedure.
- Built-In Safety Tools: Helps monitor temperature and protect healthy tissue throughout the ablation.
- Smart Scan Integration: Combines before-and-after images to plan and confirm ablation zones.
UNPARALLELED
WORKFLOW
EFFICIENCY +
VALUE
A minimally invasive approach, preferred by patients. The NeuroBlate System is the only robotically-controlled, commercially available LITT system specifically designed for use in the brain.
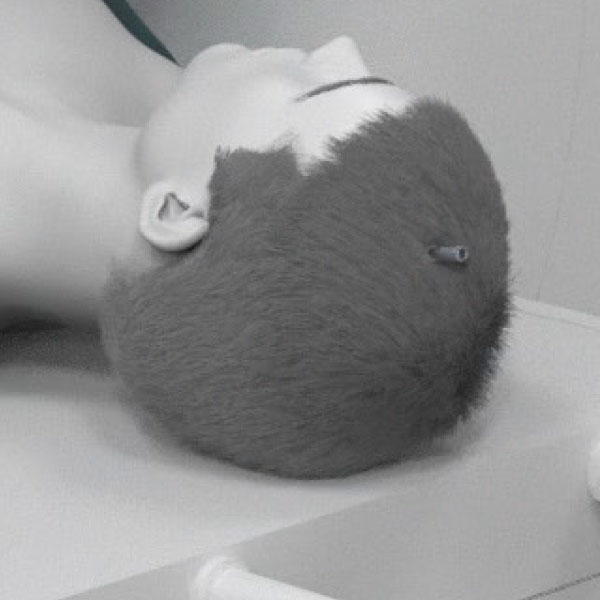
Performed through a small hole. Bolt placement in OR or interoperative MR.
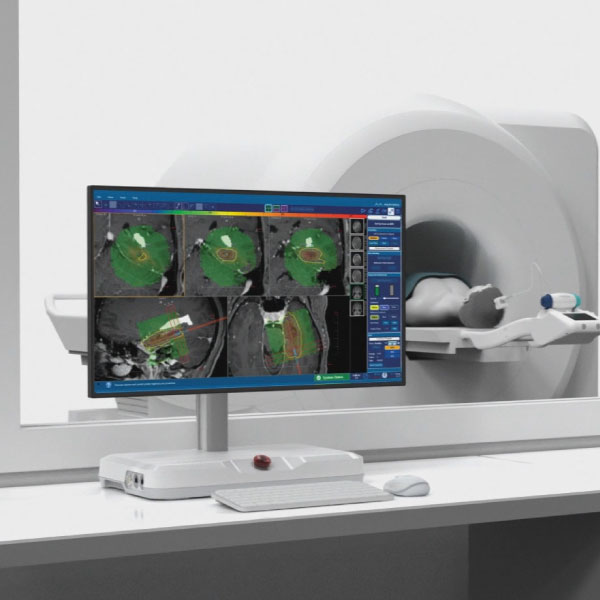
Robotic, precise manipulation of laser probe position from workstation in MR suite

Ablation is visualized and controlled remotely
Typically, patients return home after a short hospital stay with one to two stitches
Disclosures
Monteris provides technology for neurosurgeons, which allows them to ablate (destroy with heat), brain structures such as brain tumors, radiation necrosis, and epileptic foci. Monteris technology includes the NeuroBlate System, AtamA, and MiniBolt devices, which may be used together to apply the focused laser energy with little or no effect on surrounding healthy tissue. The NeuroBlate System provides clinicians a tool that offers near real-time control and MRI visualization of the therapy during laser ablation treatment.
All brain surgeries carry risk. Possible adverse events include, but are not limited to, hematoma, embolic events, edema, bleeding, cerebral spinal fluid (CSF) leakage, infection, unintended major tissue damage and permanent neurological deficits. Prior to using these devices, please review the Instructions for Use for a complete listing of indications, contraindications, warnings, precautions and potential adverse events. The intended patients are adults and pediatrics from the age of two years and older. For full prescribing information, please visit monteris.com
Rx Only