Monteris Medical Announces Congress of Neurological Surgeons Guidelines Issued on Laser Interstitial Thermal Therapy for the Treatment of Adults with Metastatic Brain Tumors
Monteris Medical Announces Market Release of NeuroBlate NB3 FullFire 1.6mm Laser Probe, Smallest Commercially Available Laser Probe for Use in the Brain
LEADING THE WAY WITH LIFE CHANGING INNOVATION.

NEW!
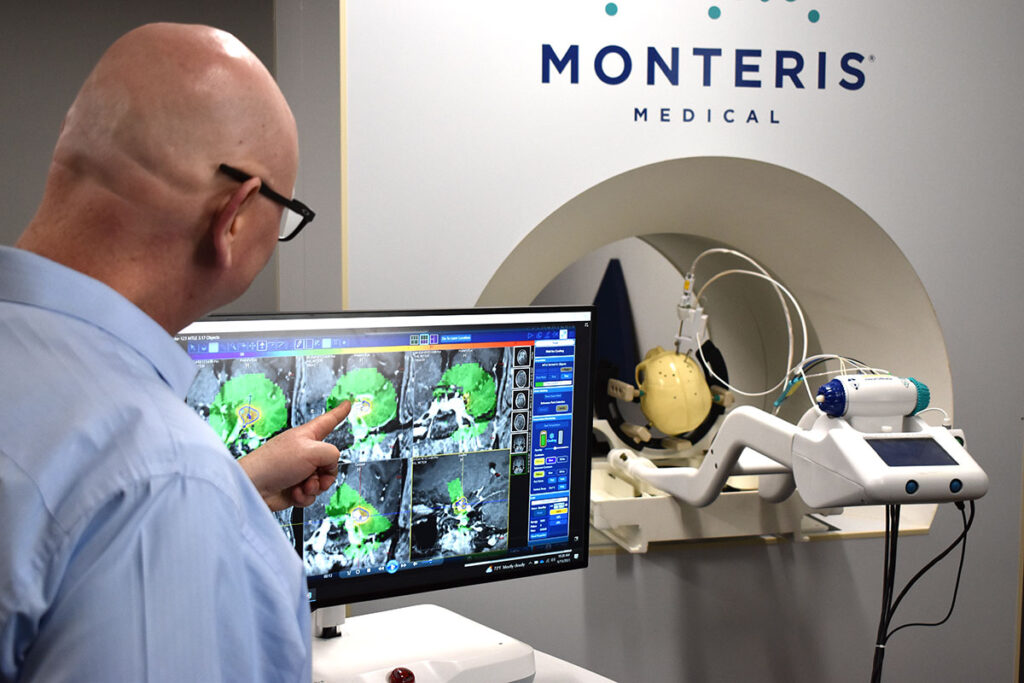
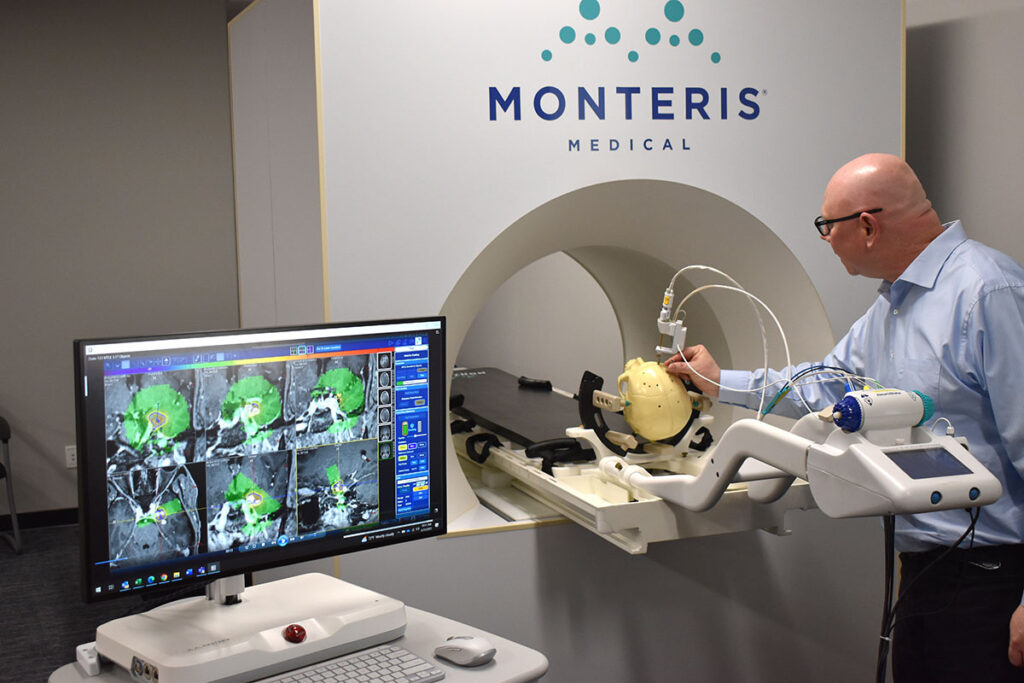
The smallest diameter, cooled laser probe designed specifically for use in the brain. NB3 ablates a wide range of lesion sizes and is available in a single length.
SUPPORTED BY EVIDENCE.
WHO WE ARE

United by our commitment to develop and deliver leading-edge neurosurgical technology.